The Booming Indian Pharmaceutical Industry: Trends, Growth Drivers, and Future Prospects
India's Pharmaceutical Industry: A Global Powerhouse
“The next big thing in
healthcare is understanding and revolutionising drug discovery through innovation and technology.- Bill
gates”
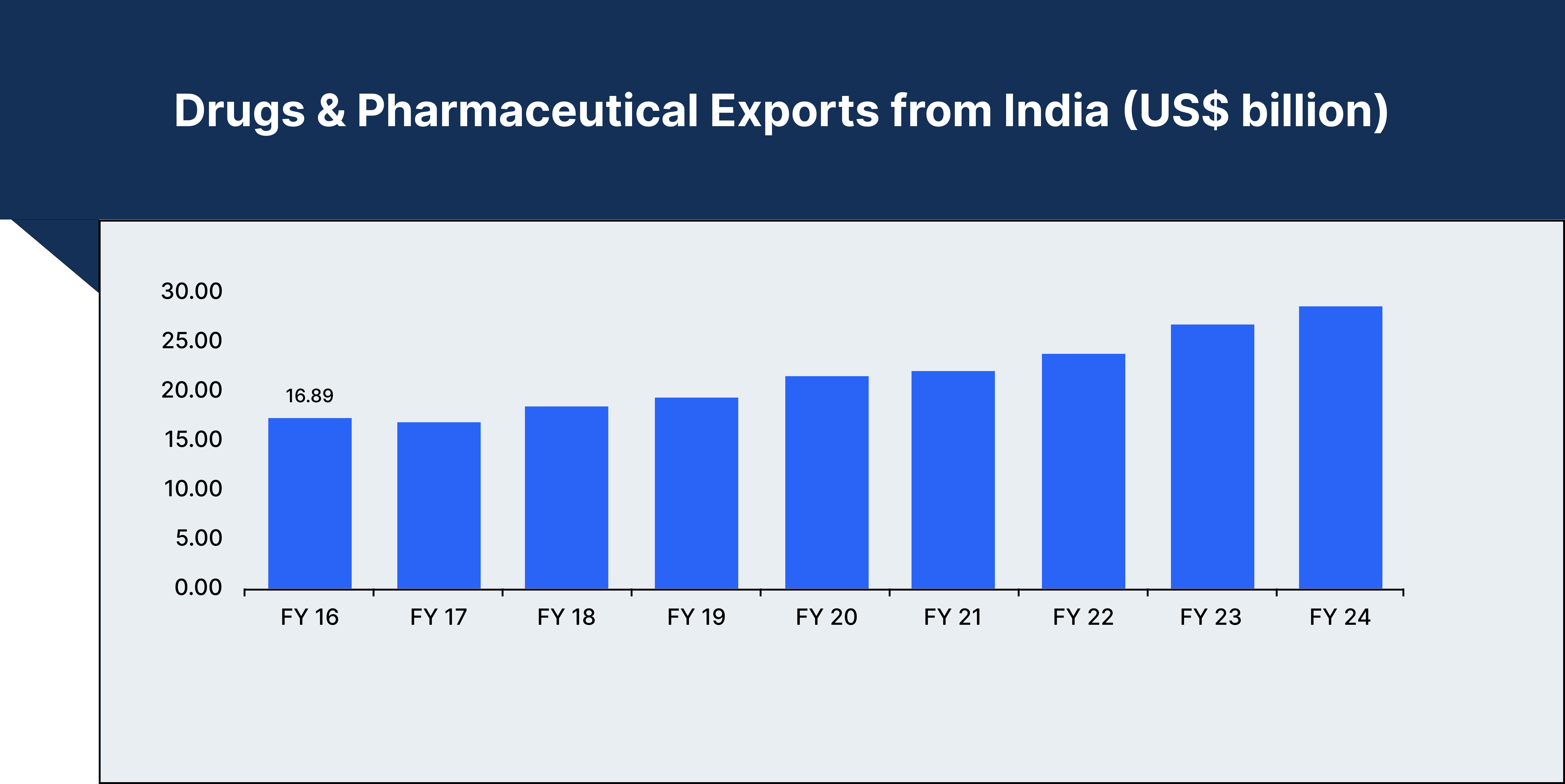
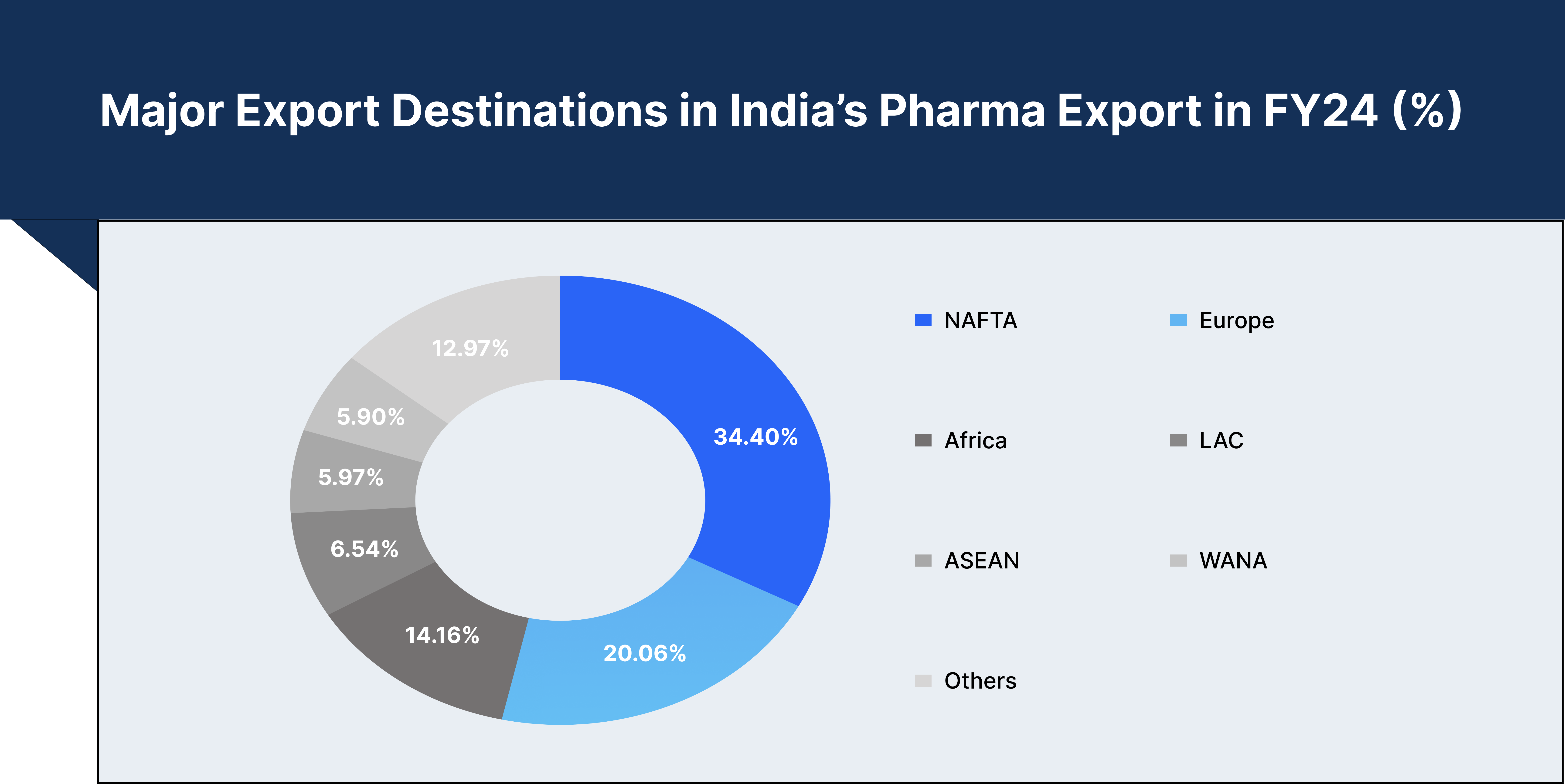
India's pharmaceutical industry is set for remarkable growth, projected to reach $65 billion by 2024, and is expected to double to $130 billion by 2030. Valued currently at $50 billion, the industry plays a pivotal role in the global supply chain, exporting medicines to more than 200 countries. India meets over 50% of Africa's demand for generic medicines, fulfils around 40% of the United States' generic drug requirements, and supplies approximately 25% of the UK's pharmaceuticals.
India also leads the world in vaccine production, accounting for nearly 60% of global vaccine demand. Major vaccines such as DPT, BCG, and measles are widely produced in the country, with 70% of WHO's essential immunisation vaccines sourced from India. This dominance in generic medicines and vaccines highlights India’s status as a key player in global healthcare.
India's pharmaceutical industry is set for remarkable growth, projected to reach $65 billion by 2024, and is expected to double to $130 billion by 2030. Valued currently at $50 billion, the industry plays a pivotal role in the global supply chain, exporting medicines to more than 200 countries. India meets over 50% of Africa's demand for generic medicines, fulfils around 40% of the United States' generic drug requirements, and supplies approximately 25% of the UK's pharmaceuticals.
India also leads the world in vaccine production, accounting for nearly 60% of global vaccine demand. Major vaccines such as DPT, BCG, and measles are widely produced in the country, with 70% of WHO's essential immunisation vaccines sourced from India. This dominance in generic medicines and vaccines highlights India’s status as a key player in global healthcare.
.png)
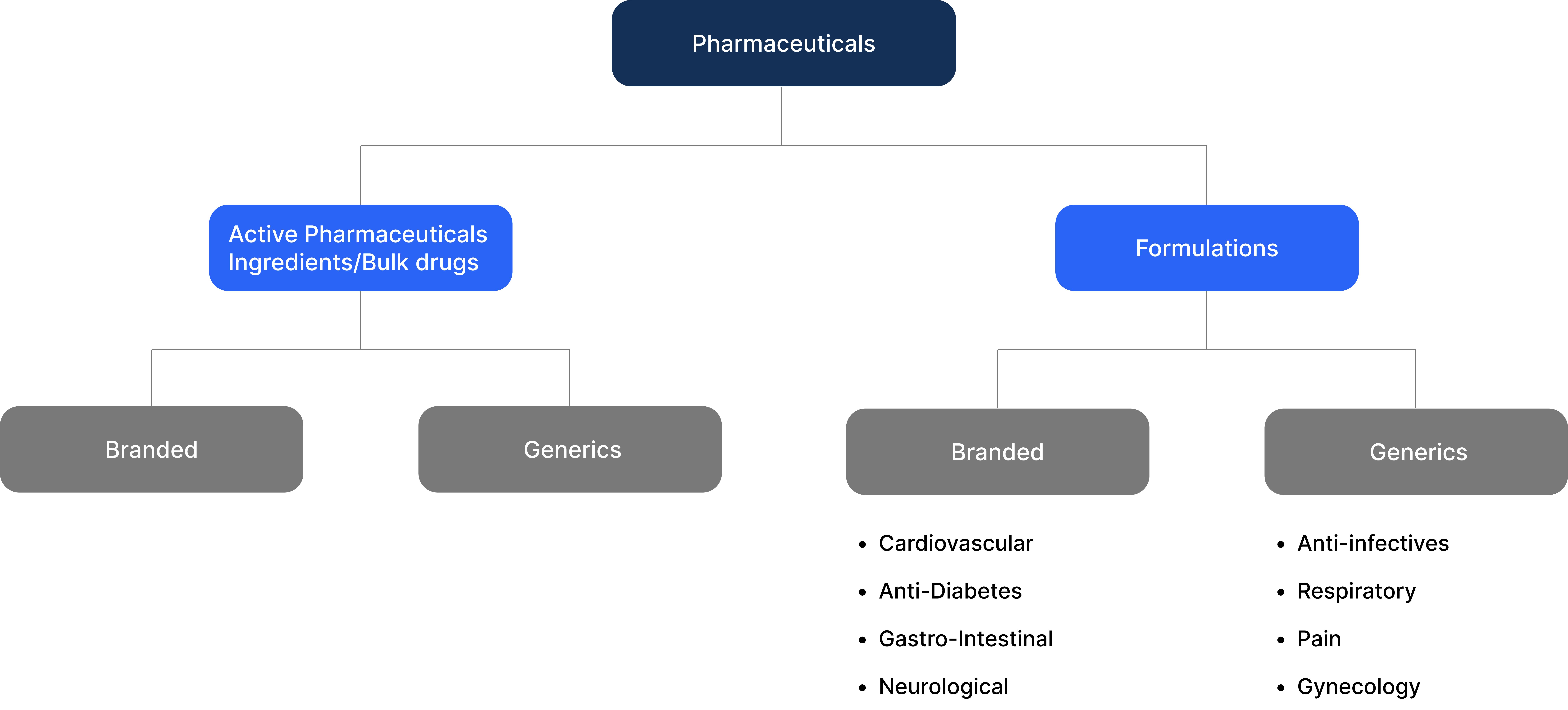
The Impact of the 1970 Patent Act
The introduction of the Patent Act in 1970 was a pivotal moment for the Indian
pharmaceutical industry. It allowed domestic companies to produce generic versions of patented drugs without
infringing on intellectual property rights, thereby reducing dependence on foreign pharmaceutical companies.
This move not only fostered the growth of the domestic industry but also positioned India as a global leader in
the production of affordable generic medicines.
Growth Drivers
Government Initiatives
Government-backed Production Linked Incentive (PLI) schemes and the establishment
of 3 bulk drug parks in Gujarat, Himachal Pradesh, & Andhra Pradesh is crucial to ensuring a steady supply
of active pharmaceutical ingredients (APIs) & enhancing India’s drug security.
Medical Tourism
India’s pharmaceutical industry benefits significantly
from the country's burgeoning medical tourism sector, which offers high-quality healthcare services at a
fraction of the cost compared to the United States, Europe, and other parts of South Asia.
Infrastructure and
Compliance
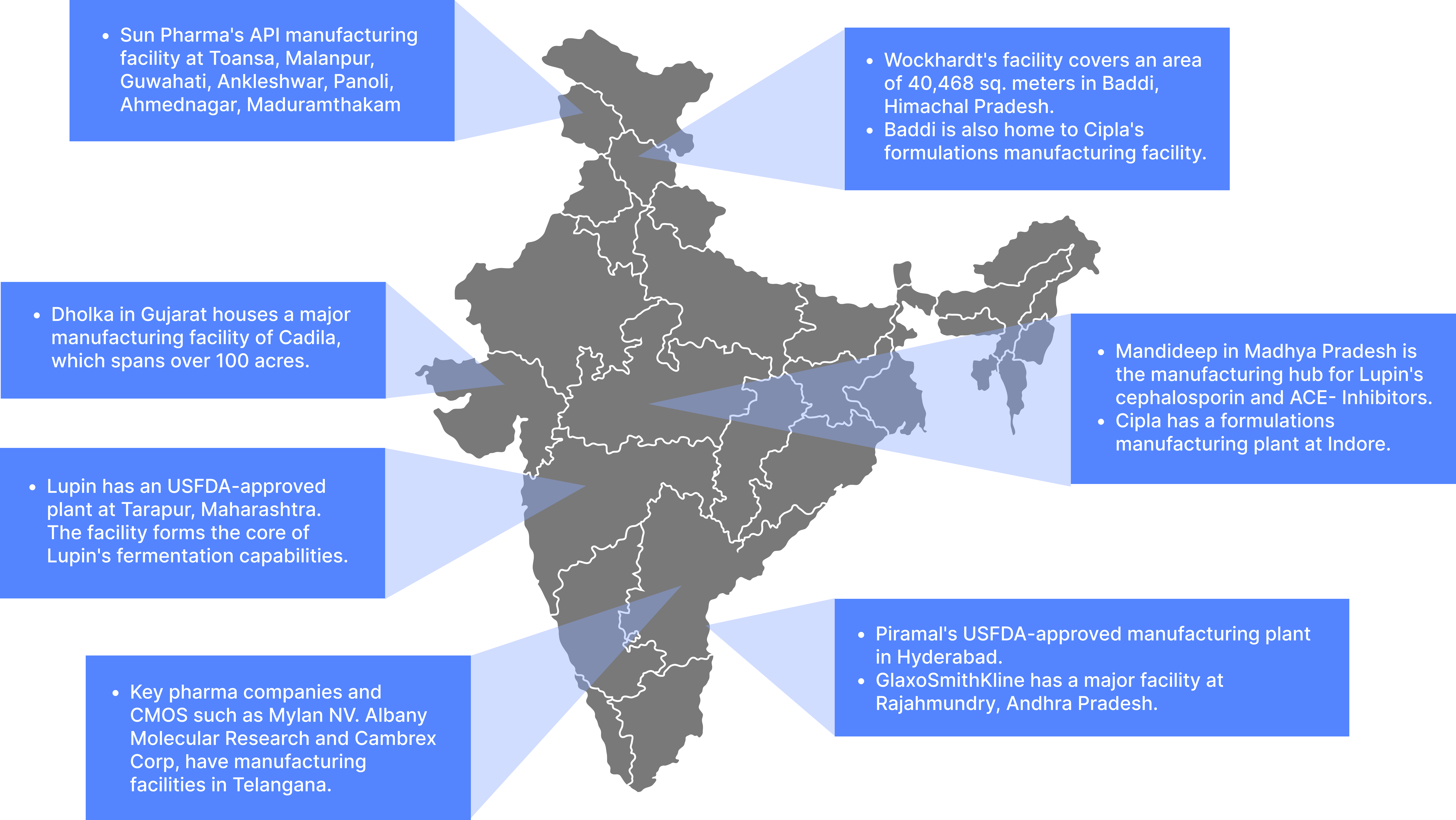
India boasts the largest number of US-FDA compliant
manufacturing plants outside of the USA,such strong infrastructure supports India’s reputation as a reliable
producer of generic drugs, including those with expired patents, and enables end-to-end drug
manufacturing.
Robust Domestic
Market
Initiatives such as the launch of the world’s largest
National Health Protection Scheme. This program aims to provide healthcare access to millions of citizens,
thereby fueling the demand for pharmaceuticals and supporting the industry’s growth.
Digital
Transformation
- Ayushman Bharat Digital Mission(ABDM): ABDM aims to create a comprehensive digital health ecosystem in India, linking patient health records to a unique Ayushman Bharat Health Account (ABHA), streamlining healthcare delivery & improving clinical decision making.
- AI and Machine Learning in Drug Development: Indian pharmaceutical companies are leveraging AI and ML to accelerate drug discovery and development processes, helping to identify potential drug candidates, optimise clinical trials, and predict patient responses.
SWOT Analysis of the Indian
Pharmaceutical Industry

The Role of the U.S. FDA in India's Pharmaceutical
Industry
The United States is a major market for Indian pharmaceutical exports, accounting for
over 25% of the total. Approximately 30% of the generic drugs sold in the U.S. are manufactured in India. As a
result, Indian pharmaceutical companies are subject to rigorous oversight by the U.S. Food and Drug
Administration (FDA), including regular site inspections and stringent quality control standards.
Interim Budget 2024-25: Pharmaceutical Sector
Highlights
In the Interim Budget 2024-25, the government allocated US$
120 million for bulk drug parks and US$ 156.5 million for pharmaceutical
development. These investments are poised to enhance India’s pharmaceutical manufacturing and research
capabilities, cementing its global position. Additionally, several Indian pharmaceutical companies benefited,
following GST reductions on cancer drugs and the introduction of the U.S. Biosecure Act,
which limits contracts with certain Chinese biotech firms. This has fueled expectations of increased U.S.
business for Indian companies.
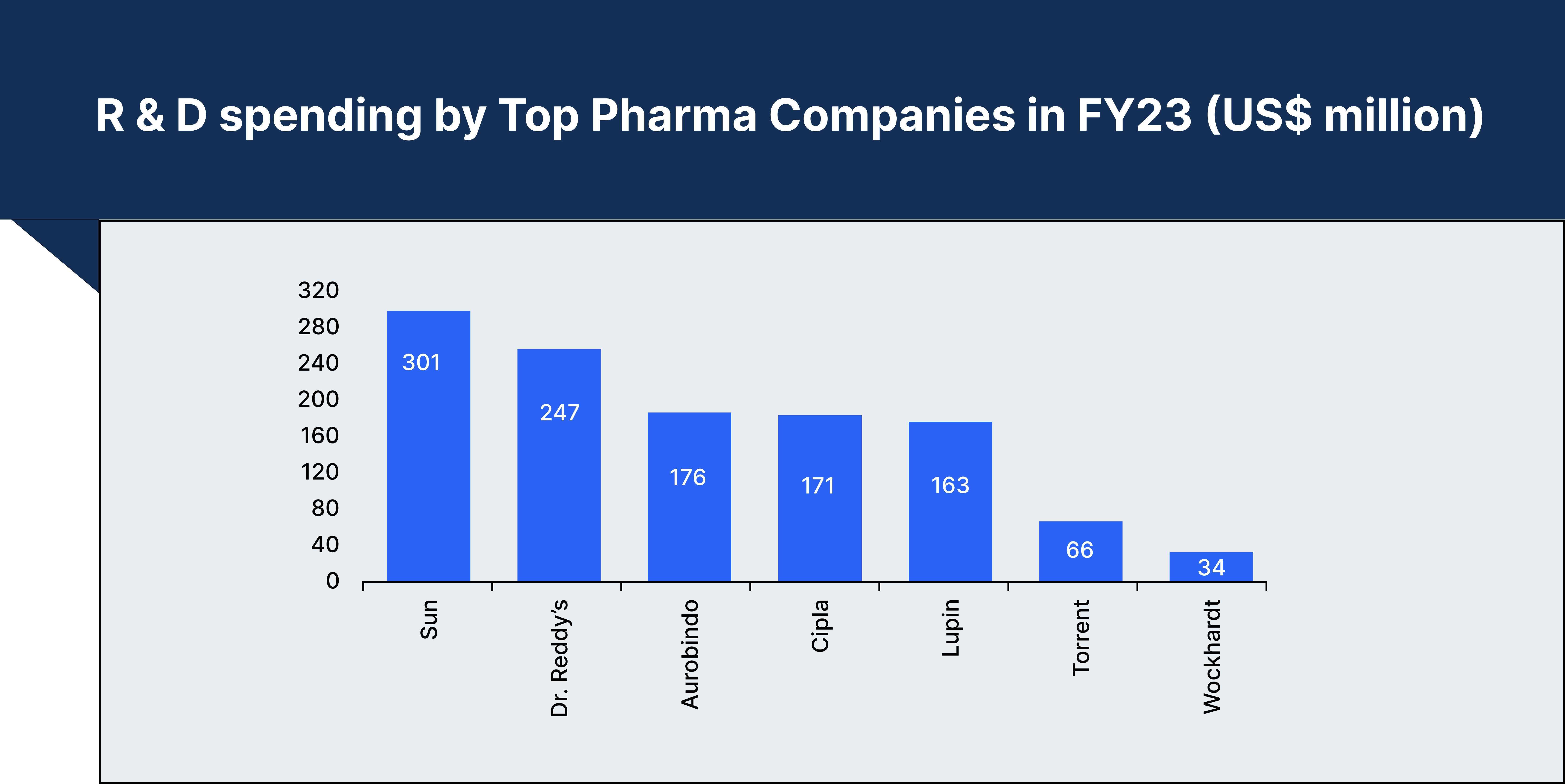
Advancements in Research &
Development
Indian pharmaceutical companies have increasingly shifted their focus towards
research and development, adapting their strategies to compete in the global market. Initially, companies
targeted generic versions of patented drugs, often challenging existing patents in court. This approach has
been sustained even after the introduction of new patent policies, with many companies now investing in the
discovery of new molecules.
The promise of high profit margins in drug discovery has attracted significant investment, with Indian companies either increasing their R&D budgets or entering into strategic partnerships to capitalise on global opportunities.
The promise of high profit margins in drug discovery has attracted significant investment, with Indian companies either increasing their R&D budgets or entering into strategic partnerships to capitalise on global opportunities.
Future Prospects for the Indian Pharmaceutical
Industry
Indian pharmaceutical companies have increasingly shifted their focus towards
research and development, adapting their strategies to compete in the global market. Initially, companies
targeted generic versions of patented drugs, often challenging existing patents in court. This approach has
been sustained even after the introduction of new patent policies, with many companies now investing in the
discovery of new molecules.
The promise of high profit margins in drug discovery has attracted significant investment, with Indian companies either increasing their R&D budgets or entering into strategic partnerships to capitalise on global opportunities.
The promise of high profit margins in drug discovery has attracted significant investment, with Indian companies either increasing their R&D budgets or entering into strategic partnerships to capitalise on global opportunities.
- Increased Focus on Biologics: There is a growing interest in biologic drugs, which are complex, large-molecule therapies derived from living organisms. Indian companies are beginning to explore opportunities in this high-growth segment.
- Digital Transformation: Adoption of AI & machine learning (ML), is expected to revolutionise drug discovery, manufacturing & distribution processes in the Indian pharmaceutical industry.
- Expansion into Emerging Markets: As developed markets become increasingly competitive, Indian pharmaceutical companies are likely to expand their presence in emerging markets, where there is rising demand for affordable healthcare solutions.
- Sustainability Initiatives: Environmental sustainability is becoming a priority for the pharmaceutical industry, as companies are adopting green manufacturing practices & reducing their carbon footprint.
The Indian pharmaceutical industry stands at a crossroads, with significant
opportunities for growth and innovation. By addressing its weaknesses and leveraging its strengths, India is
well-positioned to continue its leadership in the global pharmaceutical market.

Favourable policy measures
support growth:
- Strengthening of Pharmaceutical Industry (SPI).
- Scheme for Development of Pharma industry – Umbrella Scheme.
- Support for technology upgrades and FDIs
- Promotion of Medical Devices Parks
- Production Linked Incentive
- Biotechnology Industry Research Assistance Council
- Scheme for Promotion of Research and Innovation in Pharma MedTech Sector (PRIP)
- Production Linked Incentive (PLI)
The Indian pharmaceutical market is bolstered by several Production Linked Incentive
(PLI) Schemes designed to enhance domestic manufacturing capabilities, especially for high-value products within
the global supply chain.
- PLI Scheme for Key Starting Materials (KSMs)/Drug Intermediates (DIs) and Active Pharmaceutical Ingredients (APIs) (PLI 1.0): This initiative focuses on increasing the domestic production of 41 critical bulk drugs. Out of 51 approved projects targeting 34 specific bulk drugs, 22 had been commissioned by January 31, 2023. During this period, the scheme attracted an investment of INR 2,019 crore and created 1,900 jobs.
- PLI Scheme for Pharmaceuticals (PLI 2.0): This scheme supports pharmaceutical production, selecting 55 applicants, including 20 Micro, Small & Medium Enterprises (MSMEs). By January 31, 2023, these applicants reported sales of approximately INR 36,000 crore and invested INR 16,199 crore during the first year of implementation, generating employment for 23,000 people.

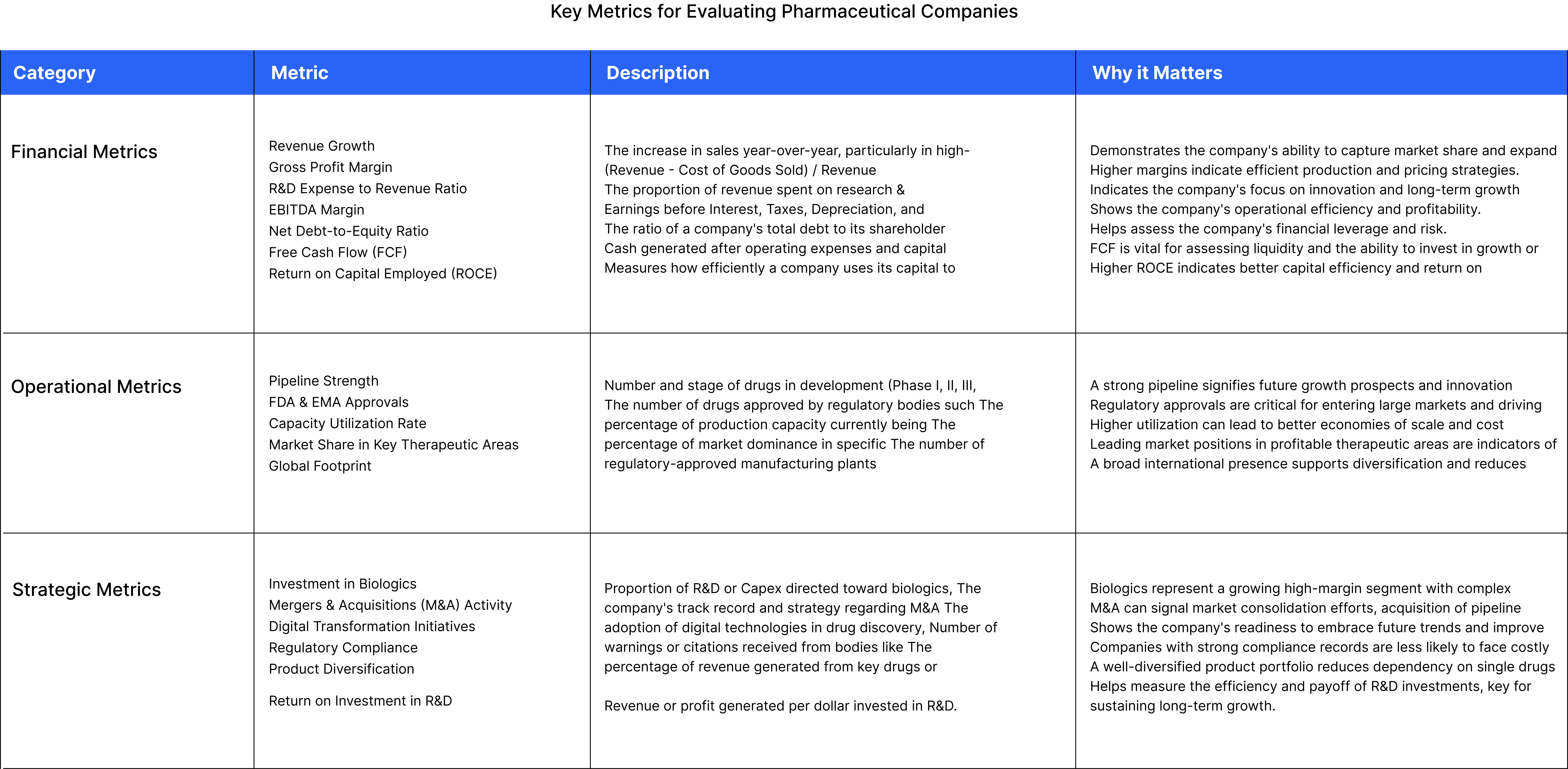
What to look out for when
evaluating Pharmaceutical Landscape